IEC 62304 vs Medical DevOps: choosing the software development process model
Software engineering has both managerial and technical aspects closely related to software life cycle.
The decision on the development process model to use in our project will have a high impact on both development and deployment
timing.
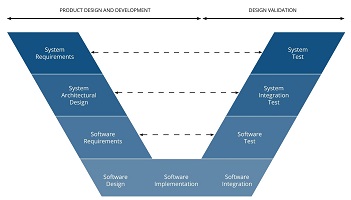
Waterfall (V) model was the first documented software life cycle model since FDA in 1985 decided that software should be taken into account when
dealing with medical devices: as IEC-62304 says, from the product requirements (namely PDP or product design proposal) as the base of development,
the V-model implements each step after the other, and does not go into the next until the latest is not verified.
If the starting requirements (PDP) are to change, as it happens daily on high-tech and research and development projects, you will need to rework all
the process.
DevOps is an incremental delivery model, that avoids some of the difficulties with V model by dividing the whole project in a set of increments.
Each increment delivers a product with somoe of the functionalities needed, so a series of loops of development and operation (namely devOp) are produced
in such a way that a partially working product exists in any step of the project.
DevOps model advantages are its flexibility and that is risk driven: the starting requirements and architecture can be redefined and verified in each loop so
the deviations from initial specifications can be managed.
Also this process matches perfectly to Agile management so each piece of the product development can be controlled, decreasing the risk while the project
progress.
 Medical DevOps en Español
Medical DevOps en Español





